Prednisone Analyzed with HPLC - AppNote
June 18, 2012
/
/
/
Isocratic Method for Prednisone Tablets
This Method uses the Mobile Phase specified in the USP Method for Assay of Prednisone tablets. The resolution between the API and the Acetanilide Internal Standard meets the system suitability.
Resolution was found to be superior compared to standard C18 HPLC Columns. In addition, this Method also produces excellent precision under these conditions, as shown in the figure overlay.
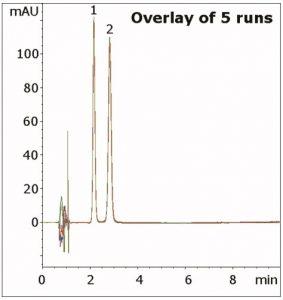

Column: Cogent Bidentate C8™, 4 µm, 100 Å
Catalog No.: 40008-75P
Dimensions: 4.6 x 75 mm
Mobile Phase: 68.8% DI Water / 25.0% Tetrahydrofuran (THF) / 6.2% Methanol (v/v)
Injection vol.: 10 µL
Flow rate: 1.0 mL / minute
Detection: UV @ 254 nm
Sample Preparation: 10 mg strength Prednisone tablet was ground and added to a 25 mL volumetric flask. It was diluted to mark with Methanol and sonicated 10 minutes. It was then filtered with a 0.45 µm Nylon Syringe Filter (MICROSOLV Technology Corp.). 110 µL of the filtrate and 90 µL of a 0.1 mg / mL Acetanilide solution were combined and diluted with 800 µL Methanol. Peaks were confirmed by individual standards.
t 0 : 0.8 minutes
Note: Prednisone is used to treat symptoms due to low corticosteroid levels. This encompasses a wide variety of applications such as for Arthritis, severe allergic reactions, Multiple Sclerosis, and Lupus. It is marketed under a variety of trade names, such as Deltasone®, Meticorten®, and Orasone®.
Attachment No 189 Prednisone Analyzed with HPLC pdf 0.5 Mb Download File
This Method uses the Mobile Phase specified in the USP Method for Assay of Prednisone tablets. The resolution between the API and the Acetanilide Internal Standard meets the system suitability.
Resolution was found to be superior compared to standard C18 HPLC Columns. In addition, this Method also produces excellent precision under these conditions, as shown in the figure overlay.


Peaks: 1. Acetanilide (Internal Standard), 2. Prednisone (API)
Method ConditionsColumn: Cogent Bidentate C8™, 4 µm, 100 Å
Catalog No.: 40008-75P
Dimensions: 4.6 x 75 mm
Mobile Phase: 68.8% DI Water / 25.0% Tetrahydrofuran (THF) / 6.2% Methanol (v/v)
Injection vol.: 10 µL
Flow rate: 1.0 mL / minute
Detection: UV @ 254 nm
Sample Preparation: 10 mg strength Prednisone tablet was ground and added to a 25 mL volumetric flask. It was diluted to mark with Methanol and sonicated 10 minutes. It was then filtered with a 0.45 µm Nylon Syringe Filter (MICROSOLV Technology Corp.). 110 µL of the filtrate and 90 µL of a 0.1 mg / mL Acetanilide solution were combined and diluted with 800 µL Methanol. Peaks were confirmed by individual standards.
t 0 : 0.8 minutes
Note: Prednisone is used to treat symptoms due to low corticosteroid levels. This encompasses a wide variety of applications such as for Arthritis, severe allergic reactions, Multiple Sclerosis, and Lupus. It is marketed under a variety of trade names, such as Deltasone®, Meticorten®, and Orasone®.
Attachment No 189 Prednisone Analyzed with HPLC pdf 0.5 Mb Download File

