Valaciclovir Analyzed with a Degradant by HPLC - AppNote
June 13, 2013
/
/
/
Separation of Prodrug from a Degradant in Forced Degradation of Valtrex® Tablets
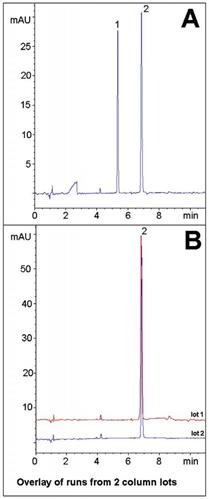
In this Application Note, the anti-viral Herpes drug Valaciclovir and its main acid degradant are well separated (Figure A). Valaciclovir is a prodrug and the degradant observed here is believed to be the active form, Acyclovir. Both compounds did not retain very strongly in Reversed Phase and the USP Method calls for a lengthy 40 minute Gradient with high water content for the Assay. The eight minute gradient provides sufficient Separation.
Data from two Column lots is shown below in the non-degraded extract (Figure B) to demonstrate the Method Reproducibility.
Column: Cogent Diamond Hydride™, 4 μm, 100 Å
Catalog No.: 70000-7.5P
Dimensions: 4.6 x 75 mm
Mobile Phase:
Post Time: 3 minutes
Injection vol.: 1 μL
Flow rate: 1.0 mL / minute
Detection: UV @ 254 nm
Sample Preparation: Stock Solution: 1000 mg strength Valtrex Tablet was ground and added to 100 mL volumetric flask containing 50 mL 50:50 DI Water / Acetonitrile diluent. The solution was sonicated 10 minutes, diluted to mark, and mixed. A portion was filtered through a 0.45 μm Nylon Syringe Filter (MICROSOLV Tech Corp.).

Attachment No 254 Valaciclovir Analyzed with HPLC pdf 0.6 Mb Download File
In this Application Note, the anti-viral Herpes drug Valaciclovir and its main acid degradant are well separated (Figure A). Valaciclovir is a prodrug and the degradant observed here is believed to be the active form, Acyclovir. Both compounds did not retain very strongly in Reversed Phase and the USP Method calls for a lengthy 40 minute Gradient with high water content for the Assay. The eight minute gradient provides sufficient Separation.
Data from two Column lots is shown below in the non-degraded extract (Figure B) to demonstrate the Method Reproducibility.


Peaks: 1. Degradant, 2. Valaciclovir
Method ConditionsColumn: Cogent Diamond Hydride™, 4 μm, 100 Å
Catalog No.: 70000-7.5P
Dimensions: 4.6 x 75 mm
Mobile Phase:
- A: DI Water with 0.1% Formic Acid (v/v)
- B: Acetonitrile with 0.1% Formic Acid (v/v)
| Time (minutes) | %B |
|---|---|
| 0 | 95 |
| 1 | 95 |
| 6 | 40 |
| 7 | 40 |
| 8 | 95 |
Post Time: 3 minutes
Injection vol.: 1 μL
Flow rate: 1.0 mL / minute
Detection: UV @ 254 nm
Sample Preparation: Stock Solution: 1000 mg strength Valtrex Tablet was ground and added to 100 mL volumetric flask containing 50 mL 50:50 DI Water / Acetonitrile diluent. The solution was sonicated 10 minutes, diluted to mark, and mixed. A portion was filtered through a 0.45 μm Nylon Syringe Filter (MICROSOLV Tech Corp.).
- Figure A: Acid Degradation Extract: The Stock Solution was diluted 1:100 with 50:50 1N HCL / Acetonitrile mixture and heated at 85°C for 30 minutes.
- Figure B: Non-Degraded Extract: The Stock Solution was diluted 1:100 with 50:50 Acetonitrile / DI Water.

Attachment No 254 Valaciclovir Analyzed with HPLC pdf 0.6 Mb Download File
