Trimethobenzamide Analyzed by HPLC - AppNote
September 4, 2012
/
/
/
Separation of Trimethobenzamide with a Simple Isocratic Method
The USP Assay Method of Trimethobenzamide is performed by titration. This alternative HPLC approach is fast, easy, and reliable. It has many advantages over the titration Method, including ease of automation and less operator-dependent results.
This simple isocratic Method provides sufficient Retention of the API to allow for Separation from other matrix components yet still is fast enough for high throughput of samples. Data from two Column lots shown in the figure illustrates the reproducibility of the Method and it robustness.
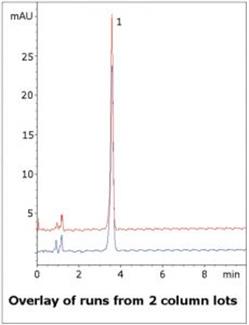
Column: Cogent Diamond Hydride™, 4 µm, 100 Å
Catalog No.: 70000-7.5P
Dimensions: 4.6 x 75 mm
Mobile Phase: 5% DI Water / 95% Acetonitrile / 0.1% (v/v) Trifluoroacetic Acid (TFA)
Injection vol.: 1 µL
Flow rate: 1.0 mL / minute
Detection: UV @ 215 nm
Sample Preparation: 1 mg Trimethobenzamide USP reference standard was dissolved in 1 mL of 50:50:0.1 (v/v) DI Water / Acetonitrile / Trifluoroacetic Acid. This stock solution was diluted 1:10 for HPLC injections using the same diluent.
t 0 : 0.9 minutes
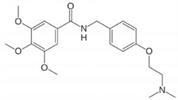
Note: Trimethobenzamide is an antiemetic used to treat nausea and vomiting that may occur after surgery or from gastroenteritis. It is sold under the trade names Tebamide® and Tigan®.
Attachment No 203 Trimethobenzamide Analyzed by HPLC pdf 0.4 Mb Download File
The USP Assay Method of Trimethobenzamide is performed by titration. This alternative HPLC approach is fast, easy, and reliable. It has many advantages over the titration Method, including ease of automation and less operator-dependent results.
This simple isocratic Method provides sufficient Retention of the API to allow for Separation from other matrix components yet still is fast enough for high throughput of samples. Data from two Column lots shown in the figure illustrates the reproducibility of the Method and it robustness.


Peak: Trimethobenzamide
Method ConditionsColumn: Cogent Diamond Hydride™, 4 µm, 100 Å
Catalog No.: 70000-7.5P
Dimensions: 4.6 x 75 mm
Mobile Phase: 5% DI Water / 95% Acetonitrile / 0.1% (v/v) Trifluoroacetic Acid (TFA)
Injection vol.: 1 µL
Flow rate: 1.0 mL / minute
Detection: UV @ 215 nm
Sample Preparation: 1 mg Trimethobenzamide USP reference standard was dissolved in 1 mL of 50:50:0.1 (v/v) DI Water / Acetonitrile / Trifluoroacetic Acid. This stock solution was diluted 1:10 for HPLC injections using the same diluent.
t 0 : 0.9 minutes
Note: Trimethobenzamide is an antiemetic used to treat nausea and vomiting that may occur after surgery or from gastroenteritis. It is sold under the trade names Tebamide® and Tigan®.
Attachment No 203 Trimethobenzamide Analyzed by HPLC pdf 0.4 Mb Download File

