Ranitidine Tablet Analyzed by HPLC - AppNote
November 8, 2012
/
/
/
Excellent Peak Shape of API From an OTC Tablet
Ranitidine has several amine functional groups that can contribute to significant tailing with Reversed Phase Methods. Data shown below uses an extract from a tablet formulation, illustrating how good Peak shape can be obtained. In addition, three Column lots were used to demonstrate the robustness and precision of this Method.
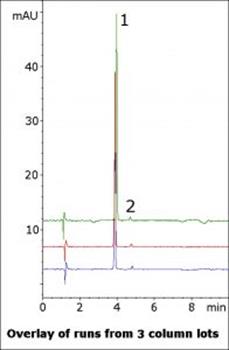
Column: Cogent Diamond Hydride™, 4 μm, 100 Å
Catalog No.: 70000-7.5P
Dimensions: 4.6 x 75 mm
Mobile Phase:
Post Time: 3 minutes
Injection vol.: 2 μL
Flow rate: 1.0 mL / minute
Detection: UV @ 313 nm
Sample Preparation: 150 mg strength Ranitidine HCl tablet was ground and weighed in a 50 mL volumetric flask. A portion of 50:50 Solvent A / Solvent B diluent was added and the flask was sonicated 10 minutes. It was then diluted to mark and filtered with a 0.45 µm Nylon Syringe Filter (MICROSOLV Tech Corp.). The filtrate was diluted 1:100 for injections.
t 0 : 0.9 minutes
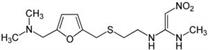
Note: Ranitidine is an acid reducer that is available over-the-counter. It works as a histamine H2-receptor antagonist, in contrast to other acid reducers such as omeprazole which are proton pump inhibitors. It is sold under the trade name Zantac®.
Attachment No 217 Ranitidine Tablet Analyzed by HPLC pdf 0.4 Mb Download File
Ranitidine has several amine functional groups that can contribute to significant tailing with Reversed Phase Methods. Data shown below uses an extract from a tablet formulation, illustrating how good Peak shape can be obtained. In addition, three Column lots were used to demonstrate the robustness and precision of this Method.


Peaks: 1. Ranitidine, 2. Matrix Component
Method ConditionsColumn: Cogent Diamond Hydride™, 4 μm, 100 Å
Catalog No.: 70000-7.5P
Dimensions: 4.6 x 75 mm
Mobile Phase:
- A: DI Water / 0.1% Trifluoroacetic acid (v/v)
- B: Acetonitrile / 0.1% Trifluoroacetic acid (v/v)
| Time (minutes) | %B |
|---|---|
| 0 | 95 |
| 1 | 95 |
| 6 | 50 |
| 7 | 95 |
Post Time: 3 minutes
Injection vol.: 2 μL
Flow rate: 1.0 mL / minute
Detection: UV @ 313 nm
Sample Preparation: 150 mg strength Ranitidine HCl tablet was ground and weighed in a 50 mL volumetric flask. A portion of 50:50 Solvent A / Solvent B diluent was added and the flask was sonicated 10 minutes. It was then diluted to mark and filtered with a 0.45 µm Nylon Syringe Filter (MICROSOLV Tech Corp.). The filtrate was diluted 1:100 for injections.
t 0 : 0.9 minutes
Note: Ranitidine is an acid reducer that is available over-the-counter. It works as a histamine H2-receptor antagonist, in contrast to other acid reducers such as omeprazole which are proton pump inhibitors. It is sold under the trade name Zantac®.
Attachment No 217 Ranitidine Tablet Analyzed by HPLC pdf 0.4 Mb Download File

