Hydromorphone Tablet Analyzed with HPLC - AppNote
May 15, 2013
/
/
/
Alternative to USP Method for this Opioid
The USP Method for Hydromorphone Tablet Assay calls for the use of Sodium Dodecyl Sulfate (SDS) in the Mobile Phase. In addition to not being LCMS compatible, adding it is time consuming to load and unload from the HPLC column. Often the columns are permanently altered and cannot be used for other analyses.
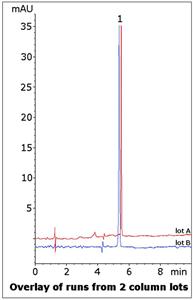
The Method conditions presented here do not require SDS and can be used in LCMS, which is advantageous since Hydromorphone is often analyzed in blood or urine matrices. Data from two column lots is shown to illustrate Method Robustness and Consistency.
Column: Cogent Diamond Hydride™, 4 μm, 100 Å
Catalog No.: 70000-7.5P
Dimensions: 4.6 x 75 mm
Mobile Phase:
-- A: DI Water with 0.1% Formic Acid (v/v)
-- B: Acetonitrile with 0.1% Formic Acid (v/v)
Gradient:
Post Time: 3 minutes
Injection vol.: 2 μL
Flow rate: 1.0 mL / minute
Detection: UV @ 280 nm
Sample Preparation: 2 mg strength Hydromorphone Tablet was ground and added to a 10 mL volumetric flask. A portion of 50:50 Solvent A / Solvent B diluent was added and the flask was sonicated 10 minutes. It was then diluted to mark and mixed. A portion was filtered with a 0.45μm Nylon Syringe Filter (MICROSOLV Tech Corp.).
t 0 : 0.9 minutes
Note: Hydromorphone is a derivative of Morphine and is used as an analgesic to treat moderate to severe pain. It is available by prescription under various trade names such as Palladone® and Dilaudid®.
Attachment No 248 Hydromorphone Tablet Analyzed with HPLC pdf 0.4 Mb Download File
The USP Method for Hydromorphone Tablet Assay calls for the use of Sodium Dodecyl Sulfate (SDS) in the Mobile Phase. In addition to not being LCMS compatible, adding it is time consuming to load and unload from the HPLC column. Often the columns are permanently altered and cannot be used for other analyses.
The Method conditions presented here do not require SDS and can be used in LCMS, which is advantageous since Hydromorphone is often analyzed in blood or urine matrices. Data from two column lots is shown to illustrate Method Robustness and Consistency.
Peak:
Hydromorphone
Column: Cogent Diamond Hydride™, 4 μm, 100 Å
Catalog No.: 70000-7.5P
Dimensions: 4.6 x 75 mm
Mobile Phase:
-- A: DI Water with 0.1% Formic Acid (v/v)
-- B: Acetonitrile with 0.1% Formic Acid (v/v)
Gradient:
| Time (minutes) | %B |
|---|---|
| 0 | 95 |
| 1 | 95 |
| 6 | 40 |
| 7 | 95 |
Post Time: 3 minutes
Injection vol.: 2 μL
Flow rate: 1.0 mL / minute
Detection: UV @ 280 nm
Sample Preparation: 2 mg strength Hydromorphone Tablet was ground and added to a 10 mL volumetric flask. A portion of 50:50 Solvent A / Solvent B diluent was added and the flask was sonicated 10 minutes. It was then diluted to mark and mixed. A portion was filtered with a 0.45μm Nylon Syringe Filter (MICROSOLV Tech Corp.).
t 0 : 0.9 minutes
Note: Hydromorphone is a derivative of Morphine and is used as an analgesic to treat moderate to severe pain. It is available by prescription under various trade names such as Palladone® and Dilaudid®.
Attachment No 248 Hydromorphone Tablet Analyzed with HPLC pdf 0.4 Mb Download File