Metronidazole Topical Cream Analyzed by HPLC - AppNote
September 18, 2013
/
/
/
Retention for Highly Polar API
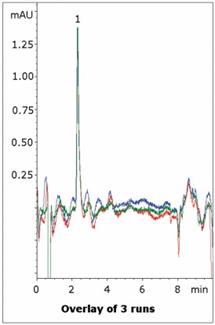
This API has very little Hydrophobic character and is not readily retained by Reversed Phase. The Peak that was obtained had excellent efficiency and symmetry. Three runs are shown in the figure in order to illustrate the repeatability of the data.
Column: Cogent Diamond Hydride™, 4 µm, 100 Å
Catalog No.: 70200-05P-2
Dimensions: 2.1 x 50 mm
Mobile Phase:
-- A: DI Water / 0.1% Formic Acid (v/v)
-- B: Acetonitrile / 0.1% Formic Acid (v/v)
Gradient:
Post Time: 5 minutes
Injection vol.: 0.2 µL
Flow rate: 0.3 mL / minute
Detection: UV @ 254 nm
Sample Preparation: 250.0 mg of 0.75% Metronidazole topical cream was added to a 25 mL volumetric flask containing a portion of 50/50 Solvent A / Solvent B diluent. After sonicating 30 minutes, it was diluted to mark. Then a portion was filtered using a 0.45µm Nylon Syringe Filter (MICROSOLV Tech Corp.).
t 0 : 0.6 minutes
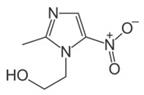
Note: Metronidazole is a Nitroimidazole antibiotic agent. It is marketed under the trade names Flagyl®, Filmet®, and others.
Attachment No 267 Metronidazole Topical Cream pdf 0.5 Mb Download File
This API has very little Hydrophobic character and is not readily retained by Reversed Phase. The Peak that was obtained had excellent efficiency and symmetry. Three runs are shown in the figure in order to illustrate the repeatability of the data.


Peak:
Metronidazole
Column: Cogent Diamond Hydride™, 4 µm, 100 Å
Catalog No.: 70200-05P-2
Dimensions: 2.1 x 50 mm
Mobile Phase:
-- A: DI Water / 0.1% Formic Acid (v/v)
-- B: Acetonitrile / 0.1% Formic Acid (v/v)
Gradient:
| Time (minutes) | %B |
|---|---|
| 0 | 95 |
| 1.5 | 95 |
| 6 | 50 |
| 7 | 95 |
Post Time: 5 minutes
Injection vol.: 0.2 µL
Flow rate: 0.3 mL / minute
Detection: UV @ 254 nm
Sample Preparation: 250.0 mg of 0.75% Metronidazole topical cream was added to a 25 mL volumetric flask containing a portion of 50/50 Solvent A / Solvent B diluent. After sonicating 30 minutes, it was diluted to mark. Then a portion was filtered using a 0.45µm Nylon Syringe Filter (MICROSOLV Tech Corp.).
t 0 : 0.6 minutes
Note: Metronidazole is a Nitroimidazole antibiotic agent. It is marketed under the trade names Flagyl®, Filmet®, and others.
Attachment No 267 Metronidazole Topical Cream pdf 0.5 Mb Download File

