Overview
This AppNote presents an effective approach for analyzing urea, a compound known for its very weak retention on conventional reversed‑phase columns due to its high polarity. The Cogent Bidentate C18™ stationary phase offers reproducible retention under ANP‑style conditions using a high‑organic mobile phase, eliminating the need for derivatization or reaction‑based sample preparation. The document outlines the chromatographic conditions, sample preparation, and advantages of this method. Additional context is provided on the analytical challenges associated with urea determination and the limitations of traditional colorimetric, enzymatic, and ion‑pairing strategies.
This method provides a cleaner, simpler, and more direct approach suitable for food, environmental, and clinical applications.
Fast and Reproducible Method for Urea Analysis Using the Cogent Bidentate C18™ Column
Urea is a highly polar compound that is notoriously difficult to retain using standard reversed‑phase HPLC columns. Under typical conditions, its retention is minimal to nonexistent. The method described here provides a simple, fast, and reproducible solution using the Cogent Bidentate C18™ column under high‑organic ANP‑type conditions.
This approach eliminates the need for derivatization or reaction steps commonly used in alternative procedures, offering a straightforward way to achieve reliable urea retention and detection.
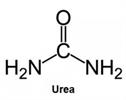
Peak:
Urea
Column: Cogent Bidentate C18™, 4μm, 100Å
Catalog No.: 40018-05P
Dimensions: 4.6 x 50mm
Mobile Phase: 15% DI Water / 85% Acetonitrile / 0.1% (v/v) Formic Acid
Flow rate: 0.2 mL /minute
Detection: UV @ 205 nm
Injection vol.: 5 μL
Sample Preparation: 3mg of Urea standard in 50:50 ACN:DI
Note: There is growing demand for a sensitive and reliable procedure for the determination of Urea in many matrices such as milk, soil extracts, seawater and wine. In addition there are several clinical applications for the analysis of this compound. The most common approach for measurement of Urea involves detection of ammonia (after hydrolysis) by color forming reactions – enzymatic, colorimetric Methods. The other techniques require noxious reagents and produce an unpleasant odor [1]. Newer Methods involve high-performance thin layer chromatography-densitometry, alkalimetric titration. HPLC is the most specific method but either organic Normal Phase Chromatography or Ion Pairing Chromatography have to be used to retain this very polar compound until this Method.
[1] “Determination of urea using HPLC with fluorescence detection after automated derivatization with xanthydrol”, S. Clark, P.S. Francis, X.A. Conlan, N.W. Barnett, J. Chromatography A, 1161 (2007) 207-213.
