Separation Science has grown to become one of the most useful scientific tools. Used extensively in diagnostic and clinical science, Separation Science is relied upon by scientists, physicians, law enforcement officials and the general public to provide quantified information about our health, our food, our environment and the products we use and in almost every aspect of our modern society.
Separation Scientists work in many different disciplines, some of these include:
- Analytical Chemistry
- Biochemistry
- Biotechnology
- Forensic Science
- Food Science
- Clinical Science
- Neuroscience
- Medical Research and Production
- Pharmaceutical and Nutraceutical Science
- Other Disciplines
Different samples and sample matrices present distinct challenges to the separation scientist which require different techniques to separate, quantify and qualify them. Smaller and smaller sample availability presents further challenges to these analyses.
Phase Partitioning and Extraction, Chromatography and Electrophoresis are the main techniques used by separation scientists today. Each technique has its own place and value in the “tool box” of those presented with the challenge of analyzing and determining the quantity of each component of a sample.
Chromatography - High Selectivity
The tool most used by scientists is Chromatography. The high selectivity of chromatography and the large body of knowledge in this field makes it the first choice of separation scientists. The mechanisms of separation make this technology ideal for small molecules such as pharmaceuticals, chemical analysis, toxicological analysis and many others. Simple automation, high reliability and robustness makes this technique highly coveted.
Electrophoresis Grows into Analytical
Electrophoresis uses a completely different mechanism of separation which offers many advantages to separation scientists. Until the advent of capillary electrophoresis (CE) and capillary electrochromatography (CEC), electrophoresis could not be automated, was not precise enough for analysis of small molecules and was limited to uses in proteins and other biomolecules.
CE and CEC have changed that as both techniques are now automated and very precise (as precise as HPLC). Robust and reliable, separation scientists now use electrophoresis as a technique for small and large molecules.
Electrophoresis is a completely different mechanism of separation from chromatography and indicated for the following reasons:
- Non Chromatographic. Provides different information and is orthogonal. Should be used with Chromatography.
- Small sample size required. Provides analysis of samples not possible before.
- Low Cost. Cost of CE and CEC is realized in the purchase and disposal costs of solvents used in Chromatography.
- Ideal for Compounds that present costly and time-consuming challenges in chromatography (proteins, peptides, nucleic acids, basic drugs and chiral compounds)
- Easy to develop methods of analysis.
What Is HPCE?
The term, “High Performance Capillary Electrophoresis”, HPCE* is an umbrella for many sub-techniques such as:
- cIEF, Capillary Iso-Electric Focusing
- MEKC, Micellar Electro-Kinetic Chromatography
- CZE, Capillary Zone Electrophoresis (also known as “Free Zone” or CE)
- CGE, Capillary Gel Electrophoresis
- CITP, Capillary Isotachophoresis
- OTCEC, Open Tubular Capillary Electrochromatography
- CEC, Capillary Electro-Chromatography
* The acronym “HPCE” (High Performance Capillary Electrophoresis) was given to Capillary Electrophoresis by chromatographers. This term is somewhat misleading as it sounds similar to HPLC but, the separation mechanism is very different. The results in HPCE may or may not be the same as in HPLC which should be considered valuable as additional sample information can be gained from this technology.
Electrophoretic separations utilize the following terms compared to its “counterpart” in HPLC.
Electrophoresis Terms
- Migration Time
- Buffer Solution/BGE
- Column Coating
- Electropherogram (e-gram)
- Theoretical Plates
- EOF, Electro Osmotic Flow
- Precision
- Efficiency
- Resolution
- Band Broadening
- Current/Voltage
Chromatography Terms
- Retention Time
- Mobile Phase
- Stationary Phase
- Chromatogram
- Theoretical Plates
- Flow Rate (loosely related)
- Precision
- Efficiency
- Resolution
- Band Broadening
- Back Pressure
Basic Theory of Capillary Electrophoresis:
Electrophoresis gets its name from the process whereby the movement of ions is produced under the influence of an applied voltage across a field that the ions exist in. It is well known that opposites attract. In electrophoresis, ions of opposite charge are attracted to and will migrate toward electrodes of opposing charge on either end of the voltage. Simply stated, ions that are negatively charged will move or migrate toward the positively charged electrode and vice versa for the positively charged ions. This is the basis for which CE and CEC operate.
Rate of Migration
What makes CE a powerful tool in separation science is the phenomena whereby every ion will migrate at different rates. This difference is based on its quantity of charge compared to its relative hydro-dynamic size. Hydro-dynamic size very closely relates to the mass of the molecule. It is commonly called the “Charge to Mass Ratio”. A simpler term that is used to describe the molecules affinity for its opposite electrode is commonly called “Electrophoretic Mobility”. These mobilities can be exploited for incredible separations of very closely structured molecules.
Ionic Mobility
The actual mobility of an ion considers the environment that the ion exists in during the separation process. For example, electrophoretic mobility will differ from actual mobility when viscosity changes and also, the amount of voltage that is applied. In simpler terms, this makes the attraction much greater or weaker to the opposite charged electrode. The more voltage, the more attraction and greater the speed.
Electro-Osmotic Flow (EOF)
CE has incredible efficiency or ability to separate similarly structured compounds due to EOF. When a voltage is applied across a tube filled with an electrolyte solution (a solution that conducts electricity) the solution begins to move toward the cathode. This mechanism is not like a chromatographic pump but, it does provide the flow of materials past a detector like an HPLC pump. This should not be confused with electrophoretic mobility described above. It is a separate phenomenon and is exploited in CE for maximum flexibility in separation power. Both EOF and Electrophoretic mobility can occur at the same time working in opposite directions to provide greater resolution.
Technical descriptions of how and why EOF occurs in a capillary is well documented and the reader of this primer should visit our web page dedicated to EOF.
Plug Flow in CE
In a capillary, the charge from electrode to electrode is conducted by the buffer system (ions in a water solution) or what is better described as the Back Ground Electrolyte (BGE). Ions of the BGE conducts electricity and provide the current needed in CE. This current is evenly distributed over the entire capillary diameter and the phenomenon of EOF occurs evenly. The water molecules in the BGE also naturally move toward the cathode in a very even manner. The electro osmotic flow that occurs is called a “plug flow” because of its plug like shape. As a result of this very even flow and flat front, extremely high Theoretical Plate Counts are observed in CE. The diagram below shows the difference between the Plug Flow of CE and the Parabolic Flow of pressure induced flow in HPLC.
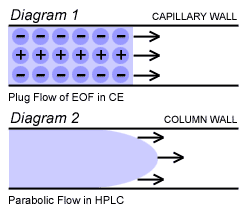
In the first diagram the EOF has a flat front due to the water molecules moving evenly toward the cathode. You can think of this in terms of them being pulled by an equal force.
In the second diagram, the parabolic flow depicts the shape of the flow due to the friction from the column walls. Pressure is applied evenly across the diameter of the column but resistance to flow occurs at the walls and a “drag” occurs. This provides a “leading edge” of separated molecules reducing the number of theoretical plates that can be achieved.
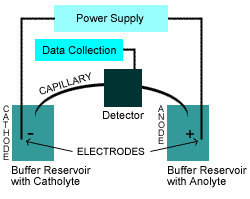
CE Instrument Schematic
A typical CE instrument uses the following components to achieve both EOF and Electrophoretic Mobility and therefore separations:
- Cathode (Negatively Charged Electrode)
- Anode (Positively Charged Electrode)
- Power Supply to generate Voltage/Current
- Catholyte (Buffer Solution at the Cathode End)
- Anolyte (Buffer Solution at the Anodic End)
- Capillary (25mm to 100mm ID)
- A Detection Method
- Data Acquisition Method
Injection
Samples are introduced into the capillary for separation by two different methods. Both having advantages and disadvantages; Electrokinetic injection and Hydrodynamic (Vacuum or Pressure) Injection.
Electrokinetic injection works when the capillary is placed into the catholyte on one end and into the anolyte (containing the sample to be analyzed) on the other end. When a voltage is applied, the EOF moves from the tip of the capillary to the end of the capillary. A siphoning effect occurs, dragging a representative sample into the capillary. Also, ions begin moving into the capillary from the buffer solution due to electrophoretic mobility as part of the sample loading. This can be an advantage when trying to analyze small concentrations of these ions. These injections usually last for 1-5 seconds.
Hydrodynamic injection works when a pressure is applied at one end of the capillary or a vacuum is applied. The pressure differential between the two opposite sides of the capillary will make liquid move into the capillary.
Temperature and therefore viscosity play a major role in reproducibility in both injections so it is important to control both.
Always Use a Water Plug
After injection, the capillary injection end is moved into a sample vial containing CE grade water. A “water plug” is injected in the same manner that the sample was injected. Then the capillary is moved into a different anolyte solution that does not contain the sample. Voltage is applied across the capillary and the separation takes place as the separated samples move (electrophoretically and with EOF) past the detector.
